Contents
Advertisement
Introduction
Definition and Etiology
Obesity has become an important public-health problem in industrialized countries throughout the world. The body mass index (BMI = weight (in kg/height2 [in m2]) is the primary measurement used to categorize obese patients (Table 1). Excess body weight (EBW) is defined as the amount of weight that is in excess of the ideal body weight (IBW). Ideal body weight is conventionally determined by the Metropolitan Life Tables, or as a BMI of 25 kg/m2. In 1991, the National Institutes of Health defined morbid obesity as a BMI of ≥ 35 kg/m2 and severe, obesity-related comorbidity as a BMI of ≥ 40 kg/m2.1
Table 1. Definitions of Obesity
Category |
Body Mass Index (kg/m2) |
Over Ideal Body Weight (%) |
| Underweight | <18.5 | |
| Normal | 18.5-24.9 | |
| Overweight | 25.0-29.9 | |
| Obesity (class 1) | 30-34.9 | >20% |
| Severe obesity (class 2) | 35-39.9 | >100% |
| Severe obesity (class 3) | 40-49.9 | |
| Superobesity | >50 | >250% |
The development of obesity involves the interactions between excessive caloric intake, inefficient use of food energy, reduced metabolic activity, a reduction in the thermogenic response to meals, and an abnormally high set point for body weight. Genetic, environmental, and psychosocial factors all contribute to this problem.
Prevalence and Risk Factors
The prevalence of obesity in the United States has increased from 15% in 1980 to 36% in 2010.2 The prevalence of extreme obesity (BMI ≥ 40 kg/m2) is 4.4% in men and 8.2% in women. The prevalence of childhood and adolescent obesity has tripled since 1980 and, currently, 17% of U.S. children and adolescents are obese.3 Obesity and morbid obesity affect women and minorities (particularly middle-aged black and Hispanic women) more than white males. However, in almost every age and ethnic group, the prevalence of overweight or obesity exceeds 50%.
Recent studies also have delineated the influence of childhood weight on adulthood weight. Being overweight during older childhood is highly predictive of adult obesity, especially if a parent also is obese. Being overweight during the adolescent years is an even greater predictor of adult obesity. Obesity is now the second leading cause of preventable death in the U.S. after cigarette smoking, despite expenditures of over $45 billion annually on weight-loss products.4
Pathophysiology and Natural History
Adipose tissue is primarily stored subcutaneously and in the abdominal cavity. In general, females are more likely to deposit fat in the peripheral tissues; males tend to deposit it in the abdominal compartment. As obesity develops, the size and number of fat cells increase. As fat cells grow, they release increasing amounts of cytokines and lower amounts of adiponectin. These changes have deleterious effects on glucose and lipid metabolism, and contribute to the proinflammatory state associated with obesity.
Obesity shortens the life span of those who suffer with it. The mortality rate of an individual with a BMI ≥ 40 kg/m2 is double that of a normal-weight individual.5 It is estimated that a man in his 20s with a BMI ≥ 45 kg/m2 has a 22% reduction in life expectancy, a decrease of 13 years.6 Most obesity-related deaths result from complications related to diabetes, cancer, and cardiovascular disease. Worldwide, approximately 2.5 million deaths occur annually because of obesity-related comorbidities.
Signs, Symptoms, and Related Diseases
There are more than 30 comorbid conditions associated with severe obesity. Insulin resistance and diabetes mellitus occur in 15% to 25% of obese patients. Increased abdominal fat in obese patients raises the intra-abdominal pressure and contributes to gastroesophageal reflux, stress urinary incontinence, venous stasis disease, and abdominal hernia. Fatty deposits in the liver can progress to nonalcoholic steatohepatitis (NASH) and, ultimately, to liver failure. Excess weight causes joint and back stress that can lead to debilitating joint disease.
Advertisement
The low-grade inflammatory state associated with morbid obesity has been implicated in the development of vascular and coronary artery disease, and the hypercoagulable state seen in these patients. Obese patients have impaired pulmonary function, particularly decreased functional residual capacity, and frequently suffer from asthma, obstructive sleep apnea, and obesity hypoventilation syndrome (also known as Pickwickian syndrome and encompassing chronic hypoxemia, hypercarbia, pulmonary hypertension, and polycythemia). Other comorbidities include hypertension, dyslipidemia, asthma, and sex-hormone dysfunction. Obesity is associated with an increased incidence of cancer of the uterus, breast, ovaries, prostate, and colon; skin infections; urinary-tract infections; migraine headaches; depression; and pseudotumor cerebri.
Diagnosis and Evaluation of Comorbidities
The diagnosis of morbid obesity is established by determining the patient’s BMI and the presence of any significant comorbid conditions. A thorough history, physical examination, and focused testing will uncover previously undiagnosed comorbidities in up to two-thirds of obese patients.
Visceral, or central, fat is more metabolically active than peripheral fat and is associated with type 2 diabetes, dyslipidemia (elevated triglyceride and reduced high-density lipoprotein [HDL] levels), high blood pressure, and increased risk for cardiovascular atherosclerotic disease. The waist-to-hip ratio helps to identify patients with excess visceral adiposity. Women with a waist-to-hip ratio >0.8 and men with a ratio >1.0 are considered to have excess central adiposity that confers risk for developing the metabolic syndrome. The diagnostic criteria for the metabolic syndrome are shown in Table 2.
Table 2. Adult Treatment Panel III Criteria for the Metabolic Syndrome*
Parameter |
Criterion |
| Central obesity | |
| Waist circumference in men | >102 cm |
| Waist circumference in women | >88 cm |
| Hypertriglyceridemia | ≥150 mg/dL |
| Low high-density lipoprotein cholesterol | |
| Men | <40 mg/dL |
| Women | <50 mg/dL |
| High blood pressure | ≥130/≥85 mmHg |
| Fasting blood glucose | ≥110 mg/dL |
*Three or more of these criteria need to be present. Adapted with permission from: National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III): Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) final report. Circulation 2002;106:3143-3421. |
|
The pretreatment evaluation performed at the Cleveland Clinic is consistent with published guidelines.7 Because obese persons are at greater risk for cardiovascular disease, a baseline electrocardiogram (ECG) should be performed. Cardiology evaluation is carried out when there is evidence of cardiac disease based on clinical symptoms or ECG findings. Chest radiography and baseline laboratory testing, including a complete blood count, chemistry panel, liver-function tests, thyroid-function tests, and a lipid profile, should be obtained as well.
Obstructive sleep apnea frequently goes unrecognized in this patient population until a thorough history prompts further evaluation. Patients with symptoms of loud snoring, daytime hypersomnolence, or a neck circumference ≥ 43 cm should undergo polysomnography and, if positive, be treated with continuous positive airway pressure (CPAP). Asthma and obesity hypoventilation syndrome also are severe pulmonary complications of obesity and should be evaluated by a pulmonologist. Dietary counseling and psychological testing are required for patients who are referred for bariatric surgery.
Treatment
Lifestyle Modifications
According to the clinical guidelines published by the American College of Physicians, all patients with a BMI ≥ 30 kg/m2 should be counseled intensively on lifestyle and behavior modifications, such as appropriate diet and exercise.8,9 An algorithm published by the American College of Physicians for medical management of an obese patient is shown in Figure 1.9 The patient’s goals for weight loss should be individually determined and may encompass other related parameters, such as decreasing blood pressure or fasting blood glucose levels. When establishing realistic weight-loss goals, it is important to realize that modest weight loss (10%-15%) is sufficient to result in health benefits.10,11
Figure 1. Algorithm for the Medical Management of Obesity
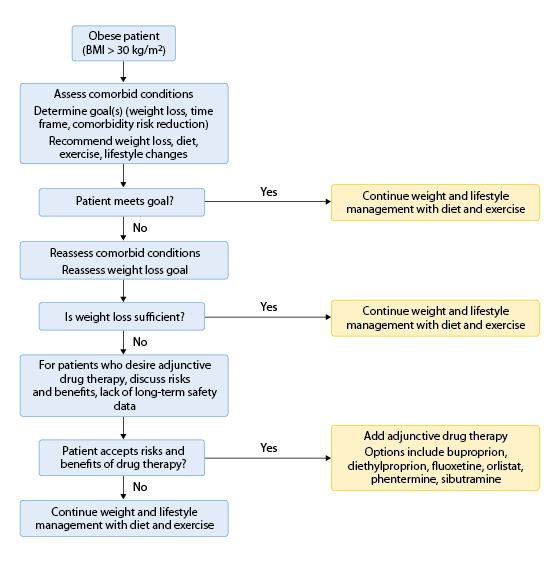
BMI, body mass index.
Adapted with permission from: Snow V, Barry P, Fitterman N, et al: Pharmacologic and surgical management of obesity in primary care: A clinical practice guideline from the American College of Physicians. Ann Intern Med 2005;142:525-531.
General dietary guidelines for achieving and maintaining a healthy weight include direction to eat a variety of nutritious foods in order to avoid vitamin deficiencies. The patient should be advised to avoid foods that are high in fat and simple sugars, and to increase dietary fiber intake. Meal-replacement shakes and behavior-modification strategies can increase adherence. The patient should be directed to maintain a diet in which 50% to 55% of calories come from complex carbohydrates. In addition, it is helpful to educate the patient about appropriate portion sizes and the caloric content of foods, as recommended by several national scientific organizations, such as the American Dietetic Association and American Diabetes Association. Referral to a registered dietitian can help the patient initiate and adhere to these dietary guidelines.
Every physician should include a graded exercise regimen as part of a comprehensive lifestyle-modification plan. Moderate exercise has been shown to decrease blood pressure; increase HDL levels and reduce triglyceride levels; and is predictive of maintenance of weight loss and delaying onset of type 2 diabetes.12 General exercise recommendations include 20 to 30 minutes of moderate exercise 5 to 7 days a week, up to 60 minutes per day most days of the week for maintenance of weight, and 90 minutes a day for achieving weight loss.
Medical Options
Pharmacologic therapy can be considered in an obese patient who has significant comorbidities or has failed to achieve weight-loss goals through lifestyle modification alone. Before initiating therapy, however, the clinician must inform the patient of any side effects associated with the drug, the lack of long-term safety data, and the temporary nature of the weight loss achieved through the use of medications. Table 3 lists the medications reviewed in the 2013 American College of Physicians clinical practice guideline for obesity management. Cardiovascular side effects were noted with the appetite suppressant sibutramine, resulting in its removal from the market.
Table 3. Medications Used for Weight Loss
Drug |
Mechanism of Action |
Side Effects |
| Phentermine/Topiramate | Appetite suppressant | Dizziness, dry mouth, constipation |
| Lorcaserin | Appetite suppressant | Nausea, dizziness |
| Phentermine | Appetite suppressant: sympathomimetic amine | Cardiovascular, gastrointestinal |
| Diethylpropion | Appetite suppressant: sympathomimetic amine | Palpitations, tachycardia, insomnia, gastrointestinal |
| Orlistat | Lipase inhibitor: decreased absorption of fat | Diarrhea, flatulence, bloating, abdominal pain, dyspepsia |
| Bupropion | Appetite suppressant: mechanism unknown | Paresthesia, insomnia, central nervous system effects |
| Fluoxetine | Appetite suppressant: selective serotonin reuptake inhibitor | Agitation, nervousness, gastrointestinal |
| Sertraline | Appetite suppressant: selective serotonin reuptake inhibitor | Agitation, nervousness, gastrointestinal |
| Topiramate | Mechanism unknown | Paresthesia, changes in taste |
| Zonisamide | Mechanism unknown | Somnolence, dizziness, nausea |
The choice of agent depends on the side-effect profile and the patient’s ability to tolerate those side effects. The amount of weight loss achieved through pharmacologic therapy is generally modest (<5 kg at 1 year). However, even modest weight loss can slow the progression of diabetes and reduce cardiovascular risk factors. However, there is no evidence that modest weight loss reduces mortality rates in these patients.
The optimal duration of treatment with obesity medications has not yet been determined. Data from randomized controlled trials are only available for up to 12 months of therapy. There are no long-term data on whether these drugs decrease morbidity or mortality from obesity-related conditions.
Surgical Options
Indications
Patients with a BMI ≥35 kg/m2 with obesity-related comorbidities, and those with a BMI >40 kg/m2 with or without comorbidities are currently eligible for bariatric surgery (Figure 2).1
Figure 2. Candidates for Bariatric Surgery
|
BMI, body mass index |
Advertisement
For a patient to be appropriate for bariatric surgery, he or she must have attempted a medical weight-loss program and should be highly motivated to make postsurgical lifestyle changes. In 1991, the National Institutes of Health (NIH) guidelines recommended that bariatric surgery be limited to patients aged 18 to 60 years. At that time, there was insufficient evidence to make recommendations about surgery for patients at the extremes of age. Although advanced age has been a predictor of increased mortality after bariatric surgery in some studies,13,14 there is evidence from case studies that bariatric surgery can be safe and effective in carefully selected adolescent and older patients.
Contraindications
Patients who cannot tolerate general anesthesia because of cardiac, pulmonary, or hepatic insufficiency are not candidates for surgery. Additionally, patients must be able to understand the consequences of the surgery and comply with the extensive preoperative evaluation and postoperative lifestyle changes, diet, vitamin supplementation, and follow-up program. Patients who have ongoing substance-abuse issues or unstable psychiatric illness are also poor candidates.
Follow-up
Bariatric surgery patients require lifetime follow-up.1 Early postoperative visits focus on potential complications or difficulties and the dramatic changes in dietary habits. Later follow-up visits focus on psychological support, nutritional assessment and vitamin supplementation, and adherence to an exercise program. Patients who present with new-onset abdominal pain, vomiting, or gastroesophageal reflux months to years after bariatric surgery should be referred to a bariatric surgeon. These symptoms may result from an anastomotic ulcer or stricture, or an intermittent bowel obstruction after Roux-en-Y gastric bypass (RYGB). Following laparoscopic adjustable gastric banding (LAGB), a new onset of gastroesophageal reflux or dysphagia may suggest gastric prolapse through the band. A patient who exhibits these signs requires prompt evaluation and treatment.
Procedures
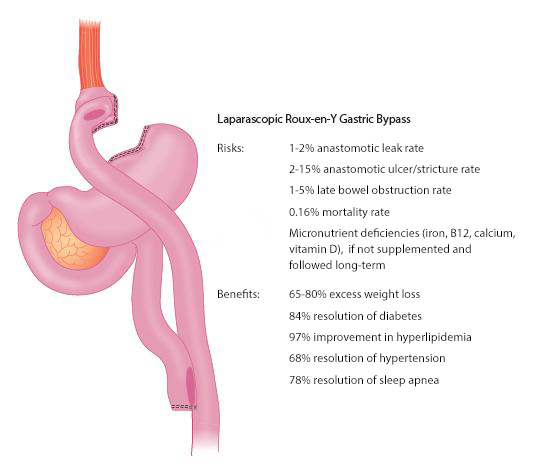
Roux-en-Y Gastric Bypass. RYGB combines a restrictive component and a limited proximal intestinal bypass, and is the most common bariatric procedure performed in the U.S. (80% of all bariatric procedures). Most RYGB procedures are now performed laparoscopically, resulting in faster recovery and fewer pulmonary and wound complications when compared with open surgery. A small, 15- to 30-mL gastric pouch is created to restrict food intake, and a Roux-en-Y anastomosis bypasses the duodenum and proximal jejunum (about 150 cm). The risks and benefits associated with RYGB are shown in Figure 3. RYGB results in superior weight loss and comorbidity resolution with excellent long-term excess weight loss (EWL) of 50% to 55% after 10 years. RYGB has unique effects on gut hormones and glucose homeostasis that are weight-loss independent. These incretin effects and a rapid improvement in diabetes have been observed in patients with mild, moderate, and severe obesity.
Figure 3. Risks and Benefits of Roux-en-Y Gastric Bypass
Laparoscopic Adjustable Gastric Banding. The first device for laparoscopic adjustable gastric banding (LAGB) was approved for use in the U.S. in 2001 after demonstrating excellent results in Europe and Australia. In this procedure, a silicone band with an inflatable inner collar is placed around the upper portion of the stomach to create a small gastric pouch and to restrict the gastric cardia. The band is connected to a port that is placed in the subcutaneous tissue of the abdominal wall. The inner diameter of the band can be adjusted by injecting saline through the port.
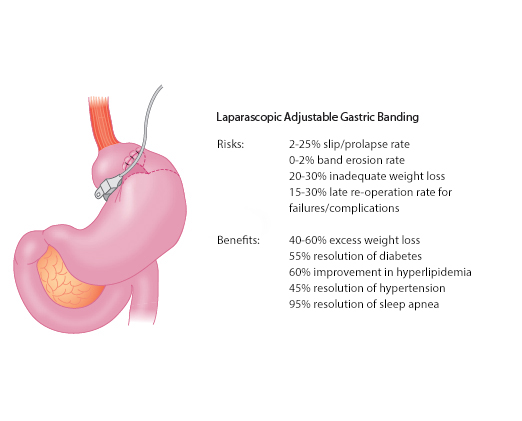
The adjustability of LAGB is a major advantage over vertical-banded gastroplasty. Band adjustments are made according to weight loss, hunger, and satiety by injecting or removing saline via the subcutaneous port. Severe complications and mortality rates are lower for LAGB than for RYGB, but LAGB typically results in less weight loss that occurs more gradually. Common risks and benefits of LAGB are shown in Figure 4.
Figure 4. Risks and Benefits of Laparoscopic Adjustable Gastric Banding
Sleeve Gastrectomy. Laparoscopic sleeve gastrectomy (LSG) has been used as a weight-loss procedure in a variety of patient groups for more than 10 years. The risks and benefits of this procedure are described in Figure 5. This operation involves a vertical resection and removal of the body and fundus of the stomach, leaving a tubular gastric lumen from the gastroesophageal junction to the antrum. The pylorus is left intact and there is no device or bypass associated with this procedure. Initially, LSG was used as part of a risk-management strategy for high-risk and very high BMI patients. After achieving substantial weight loss and improved health status following LSG, these patients underwent an RYGB or duodenal switch procedure to continue the weight loss. In the past several years, LSG has gained acceptance as a primary bariatric procedure for lower BMI patients, as well. Early complications include leaks at the gastric staple line in 1% to 2% of patients, bleeding, and strictures or narrowing at the gastric incisura. The re-operation and late complication rates for LSG are lower than for LAGB and RYGB, but overall LSG has been shown to fall between LAGB and RYGB in terms of risks, benefits, and weight loss.
Figure 5. Risks and Benefits of Laparoscopic Sleeve Gastrectomy
| Risks: |
|
| Benefits: |
|
Biliopancreatic Diversion. Biliopancreatic diversion is a malabsorptive procedure performed by less than 3% of bariatric surgeons in the U.S. This procedure is designed to limit intestinal energy absorption to the length of the distal common channel. Although these procedures offer the best and most durable weight-loss results of any bariatric procedure performed today, higher complication rates, nutritional deficiencies, and a higher mortality rate have limited their widespread use.
Advertisement
Outcomes
In a meta-analysis of 29 studies of the drug orlistat (also called tetrahydrolipstatin), the pooled mean weight loss for orlistat-treated patients was 2.59 kg at 6 months and 2.89 kg at 12 months. The average age of patients enrolled was 48 years, and the average BMI was 36.7 kg/m2.15
Other agents—such as phentermine, diethylpropion, and fluoxetine—result in a 3.0- to 3.6-kg weight loss after 1 year when used in combination with lifestyle modifications. There are limited data on sertraline, bupropion, topiramate, and zonisamide with regard to weight-loss outcomes. Therefore, recommendations cannot be made until further studies have been completed.16
A randomized, controlled trial evaluating the use of the laparoscopic adjustable gastric band (lap-band) for mild to moderate obesity (BMI 30 kg/m2 to 35 kg/m2) has demonstrated significantly greater weight loss and comorbidity resolution among patients in the surgical group compared with those enrolled in an aggressive medical weight-loss program. After 2 years, EWL was 87% in the surgical group and 21% in the nonsurgical group. Metabolic syndrome resolved in 93% of surgical patients and in 47% of nonsurgical patients.17
A large, prospective, matched cohort study (Swedish Obese Subjects Study) demonstrated the durability of weight loss and comorbidity reduction 10 years after bariatric surgery18 and a 30% reduction in all-cause mortality in the surgery group.19 Another large, matched cohort study demonstrated a significant reduction in mortality (40% reduction in all-cause mortality) 7 years after gastric-bypass surgery.20
A meta-analysis analyzing 22,094 patients in 136 studies found that for all bariatric procedures, the average EWL was 61.2%. Biliopancreatic diversion or duodenal switch procedures had the highest overall EWL (70%), followed by gastric bypass (61%), and gastric banding (47%). Overall, diabetes improved or resolved in 86% of patients, hyperlipidemia improved in 70%, hypertension improved or resolved in 78.5%, and obstructive sleep apnea improved or resolved in 83.6% of patients.21 Two recent randomized controlled trials have demonstrated the efficacy of bariatric surgery over medical therapy for the treatment of type 2 diabetes, often eliminating the need for hypoglycemic and cardiovascular drugs.22,23
In a meta-analysis of more than 85,000 patients, the overall postoperative mortality after bariatric surgery (<30 days) was 0.3%. The operative mortality rates for laparoscopic restrictive procedures and laparoscopic gastric bypass were 0.07% and 0.16%, respectively.24 Mortality after bariatric surgery is primarily the result of pulmonary embolism, anastomotic leak, or septic complications.
Summary
- Severe obesity can adversely affect every organ system.
- Detailed evaluation of symptoms can uncover serious comorbidities.
- Diabetes, cancer, and cardiovascular disease are common in obese patients.
- Central adiposity is associated with the presence of the metabolic syndrome.
- All obese patients (BMI >30 kg/m2) should be counseled on lifestyle and behavioral modifications, such as appropriate diet and exercise.
- Pharmacologic therapy can be offered to obese patients who have failed to lose weight through exercise and changes in diet.
- Bariatric surgery should be considered for morbidly obese patients in whom medical weight-loss programs─diet and exercise, with or without pharmacotherapy─have failed. Patients with a BMI >40 kg/m2, or >35 kg/m2 with obesity-related comorbidities, are candidates for bariatric surgery.
- Bariatric surgery should be considered for the treatment of type 2 diabetes in severely and moderately obese individuals.
Suggested Readings & References
Suggested Readings
- Arterburn DE, Crane PK, Veenstra DL: The efficacy and safety of sibutramine for weight loss: A systematic review. Arch Intern Med 2004;164:994-1003.
- Buchwald H, Avidor Y, Braunwald E, et al: Bariatric surgery: A systematic review and meta-analysis. JAMA 2004;292:1724-1737.
- Christou NV, Sampalis JS, Liberman M, et al: Surgery decreases long-term mortality, morbidity, and health care use in morbidly obese patients. Ann Surg 2004;240:416-423.
- Consensus Development Conference Panel: NIH conference. Gastrointestinal surgery for severe obesity. Ann Intern Med 1991;115:956-961.
- Demaria EJ, Jamal MK: Surgical options for obesity. Gastroenterol Clin North Am 2005;34:127-142.
- Flegal KM, Graubard BI, Williamson DF, Gail MH: Excess deaths associated with underweight, overweight, and obesity. JAMA 2005;293:1861-1867.
- Flum DR, Salem L, Elrod JA, et al: Early mortality among Medicare beneficiaries undergoing bariatric surgical procedures. JAMA 2005;294:1903-1908.
- Fontaine KR, Redden DT, Wang C, et al: Years of life lost due to obesity. JAMA 2003;289:187-193.
- Haddock CK, Poston WS, Dill PL, et al: Pharmacotherapy for obesity: A quantitative analysis of four decades of published randomized clinical trials. Int J Obes Relat Metab Disord 2002;26:262-273.
- Harris MI, Flegal KM, Cowie CC, et al: Prevalence of diabetes, impaired fasting glucose, and impaired glucose tolerance in U.S. adults. The Third National Health and Nutrition Examination Survey, 1988-1994. Diabetes Care 1998;21:518-524.
- Knowler WC, Barrett-Connor E, Fowler SE, et al: Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med 2002;346:393-403.
- Livingston EH, Huerta S, Arthur D, et al: Male gender is a predictor of morbidity and age a predictor of mortality for patients undergoing gastric bypass surgery. Ann Surg 2002;236:576-582.
- McTigue KM, Harris R, Hemphill B, et al: Screening and interventions for obesity in adults: Summary of the evidence for the U.S. Preventive Services Task Force. Ann Intern Med 2003;139:933-949.
- National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III): Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) final report. Circulation 2002;106:3143-3421.
- Ogden CL, Carroll MD, Curtin LR, et al: Prevalence of overweight and obesity in the United States, 1999-2004. JAMA 2006;295:1549-1555.
- Pi-Sunyer FX. A review of long-term studies evaluating the efficacy of weight loss in ameliorating disorders associated with obesity. Clin Ther 1996;18:1006-1035.
- Sauerland S, Angrisani L, Belachew M, et al: Obesity surgery: Evidence-based guidelines of the European Association for Endoscopic Surgery (EAES). Surg Endosc 2005;19:200-221.
- Sjostrom L, Lindroos AK, Peltonen M, et al: Lifestyle, diabetes, and cardiovascular risk factors 10 years after bariatric surgery. N Engl J Med 2004;351:2683-2693.
- Snow V, Barry P, Fitterman N, et al: Pharmacologic and surgical management of obesity in primary care: A clinical practice guideline from the American College of Physicians. Ann Intern Med 2005;142:525-531.
- Wadden TA, Bartlett SJ, Foster GD, et al: Sertraline and relapse prevention training following treatment by very-low-calorie diet: A controlled clinical trial. Obes Res 1995;3:549-557.
- Wolf AM, Colditz GA: The cost of obesity: The U.S. perspective. Pharmacoeconomics 1994;5(Suppl 1):34-37.
References
- NIH conference. Gastrointestinal surgery for severe obesity. Consensus Development Conference Panel. Ann Intern Med 1991; 115(12):956-61.
- Flegal KM, Carroll MD, Kit BK, Ogden CL. Prevalence of obesity and trends in the distribution of body mass index among US adults, 1999-2010. Jama; 307(5):491-7.0
- Ogden CL, Carroll MD, Kit BK, Flegal KM. Prevalence of obesity and trends in body mass index among US children and adolescents, 1999-2010. Jama; 307(5):483-90.
- Wolf AM, Colditz GA. The cost of obesity: the US perspective. Pharmacoeconomics 1994; 5(Suppl 1):34-7.
- Flegal KM, Graubard BI, Williamson DF, Gail MH. Excess deaths associated with underweight, overweight, and obesity. Jama 2005; 293(15):1861-7.
- Fontaine KR, Redden DT, Wang C, et al. Years of life lost due to obesity. Jama 2003; 289(2):187-93.
- Sauerland S, Angrisani L, Belachew M, et al. Obesity surgery: evidence-based guidelines of the European Association for Endoscopic Surgery (EAES). Surg Endosc 2005; 19(2):200-21.
- Snow V, Barry P, Fitterman N, et al. Pharmacologic and surgical management of obesity in primary care: a clinical practice guideline from the American College of Physicians. Ann Intern Med 2005; 142(7):525-31.
- McTigue KM, Harris R, Hemphill B, et al. Screening and interventions for obesity in adults: summary of the evidence for the U.S. Preventive Services Task Force. Ann Intern Med 2003; 139(11):933-49.
- Pi-Sunyer FX. A review of long-term studies evaluating the efficacy of weight loss in ameliorating disorders associated with obesity. Clin Ther 1996; 18(6):1006-35; discussion 1005.
- Harris MI, Flegal KM, Cowie CC, et al. Prevalence of diabetes, impaired fasting glucose, and impaired glucose tolerance in U.S. adults. The Third National Health and Nutrition Examination Survey, 1988-1994. Diabetes Care 1998; 21(4):518-24.
- Knowler WC, Barrett-Connor E, Fowler SE, et al. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med 2002; 346(6):393-403.
- Flum DR, Salem L, Elrod JA, et al. Early mortality among Medicare beneficiaries undergoing bariatric surgical procedures. Jama 2005; 294(15):1903-8.
- Livingston EH, Huerta S, Arthur D, et al. Male gender is a predictor of morbidity and age a predictor of mortality for patients undergoing gastric bypass surgery. Ann Surg 2002; 236(5):576-82.
- Haddock CK, Poston WS, Dill PL, et al. Pharmacotherapy for obesity: a quantitative analysis of four decades of published randomized clinical trials. Int J Obes Relat Metab Disord 2002; 26(2):262-73.
- Wadden TA, Bartlett SJ, Foster GD, et al. Sertraline and relapse prevention training following treatment by very-low-calorie diet: a controlled clinical trial. Obes Res 1995; 3(6):549-57.
- O’Brien PE, Dixon JB, Laurie C, et al. Treatment of mild to moderate obesity with laparoscopic adjustable gastric banding or an intensive medical program: a randomized trial. Ann Intern Med 2006; 144(9):625-33.
- Sjostrom L, Lindroos AK, Peltonen M, et al. Lifestyle, diabetes, and cardiovascular risk factors 10 years after bariatric surgery. N Engl J Med 2004; 351(26):2683-93.
- Sjostrom L, Narbro K, Sjostrom CD, et al. Effects of bariatric surgery on mortality in Swedish obese subjects. N Engl J Med 2007; 357(8):741-52.
- Adams TD, Gress RE, Smith SC, et al. Long-term mortality after gastric bypass surgery. N Engl J Med 2007; 357(8):753-61.
- Buchwald H, Avidor Y, Braunwald E, et al. Bariatric surgery: a systematic review and meta-analysis. Jama 2004; 292(14):1724-37.
- Mingrone G, Panunzi S, De Gaetano A, et al. Bariatric surgery versus conventional medical therapy for type 2 diabetes. N Engl J Med; 366(17):1577-85.
- Schauer PR, Kashyap SR, Wolski K, et al. Bariatric surgery versus intensive medical therapy in obese patients with diabetes. N Engl J Med; 366(17):1567-76.
- Buchwald H, Estok R, Fahrbach K, et al. Trends in mortality in bariatric surgery: a systematic review and meta-analysis. Surgery 2007; 142(4):621-32; discussion 632-5.
